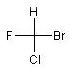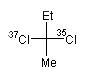| The R and S stereodescriptors are IUPAC-approved designations of absolute configuration at four-coordinate (quadriligant) and six-coordinate (sexiligant) stereogenic centers [1]. In CurlySMILES an R/S enantiomer is encoded by anchoring the corresponding curly codes {R} and {S} at stereogenic centers of a chiral molecule. The following examples illustrates this for two chiral molecules (presented as Fischer projections) with one asymmetric C-atom: | ||||||
|
||||||
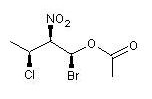
| A molecules with multiple asymmetric atoms is encoded in the same way by anchoring the respective stereodescriptors at the stereogenic-center atoms: | ||||||
|
||||||
Tweet
_
__
__
 __
__
__ ___ 
|
References
| [1] | G. P. Moss (prepared recommondations for publication): Basic Terminology of Stereochemistry. Pure & Appl. Chem. 1996, 68 (12), pp. 2193-2222. pdf: www.iupac.org/publications/pac/1996/pdf/6812x2193.pdf . |
| [2] |
B. Testa:
Principles of organic stereochemistry. Marcel Dekker, New York and Basel, 1979. |
| [3] |
D. Hellwinkel:
Systematic Nomenclature of Organic Chemistry. Springer-Verlag, Berlin und Heidelberg, Germany, 2001. |
| [4] | A. Drefahl: CurlySMILES: a chemical language to customize and annotate encodings of molecular and nanodevice structures. J. Cheminf. 2011, 3:1; doi: 10.1186/1758-2946-3-1 . |
| Encoding of stereoisomers |
D and L
enantiomersE and Z isomers |

Custom Search
|